NAP TECHNOLOGY
What are nucleic acid polymers?
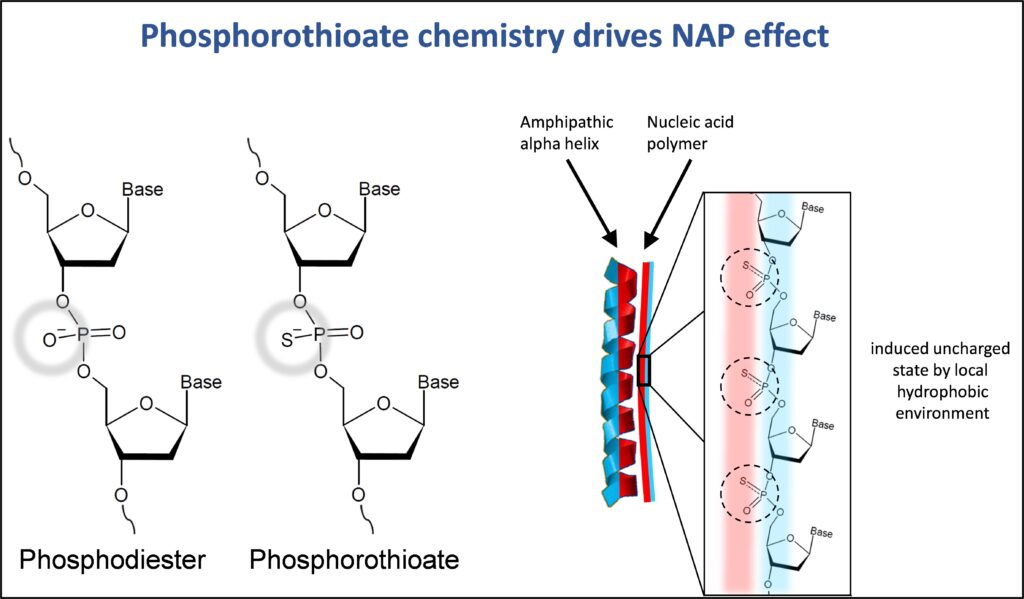
Nucleic acid polymers are oligonucleotides which exert their biological effects by the sequence independent
properties of oligonucleotides. NAP polymers containing the phosphorothioate modification (where the nonbridging oxygen is replaced with sulfur) have an ability to anneal along the exposed hydrophobic surface of
exposed amphipathic alpha helices and block conformational changes.
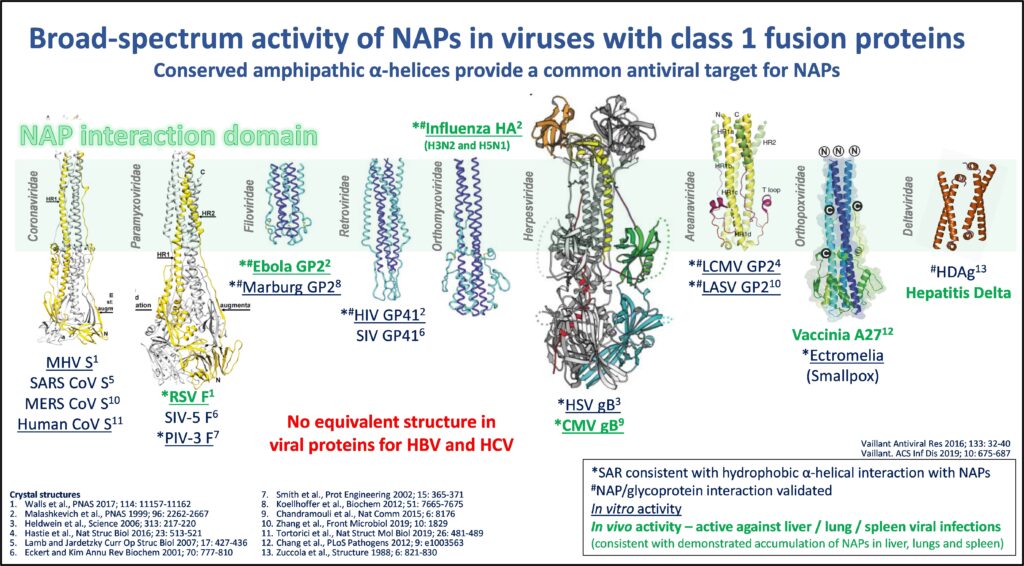
While these exposed hydrophobic protein domains are rare in mammalian biology, they are quite common in
viral systems, leading to a broad-spectrum antiviral effect in a variety of enveloped viruses.
As antiviral agents, REP 2139-based therapy will not result in the development of resistant strains of virus due
to the large binding interface between NAPs and the target protein, which is insensitive to mutation.
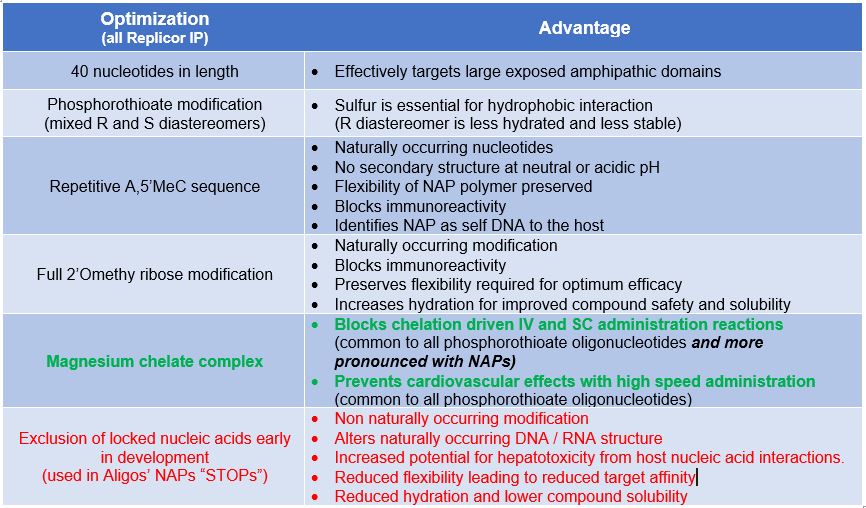
The optimized NAP, REP 2139
The biological activity of NAPs is sequence independent and also not affected by certain modifications (such
as modifications to the ribose sugar). Replicor has taken advantage of this flexibility to engineer its lead NAP
compound, REP 2139, with a proprietary, ribose modified, poly adenosine-cytidine sequence containing
naturally occurring nucleotide modifications. These modifications allow REP 2139 to retain activity but without
causing off target effects (such as immunoreactivity or hepatoxicity) which results in it being very safe and well
tolerated.
REP 2139-Mg is the lead clinical NAP drug product which has undergone several optimizations essential for
its excellent efficacy, tolerability and safety: