Hepatitis B
There are an estimated 292 million patients with chronic hepatitis B (HBV) infection worldwide. More than 880,000 people die from complications directly related to their chronic HBV infection every year. Current therapies can provide control of the virus but suffer from an inability to remove antigen burden (HBsAg) from patients, an essential step required to restore functional control of infection, which remains after current treatments are withdrawn.
What is Hepatitis B?
The hepatitis B virus (HBV) infects the liver and causes inflammation (hepatitis). More than 2 billion people
worldwide have been infected with HBV, leaving an estimated 292 million people with chronic HBV infection.
In the vast majority of people who have resolved their infection, it is actually still present in the liver but in a
latent state because of effective and continual control by their immune system. In patients who cannot
establish this immune control, HBV infection becomes chronically active. In individuals who resolved their
infection but subsequently receive immunosuppressive therapy (even decades later), HBV can reactivate.
In patients with chronically active HBV infection, liver inflammation leads to fibrosis and cirrhosis. In addition,
progressive integration of viral DNA in host chromosomes can lead to the onset of liver cancer. As a result,
approximately 880,000 patients with chronic HBV infection will eventually die each year from these
complications due to chronic HBV infection. For more information on HBV, please visit the World Health
Organization website here.
Clearance of HBV subviral particles – a critical hurdle in achieving functional cure of HBV
Patients who achieve immunological control of their HBV infection in the absence of therapy are said to have
entered a state of “functional cure”, which is characterized by levels of the viral antigen known as hepatitis B
surface protein (or HBsAg) below the limit of quantification of current assays, undetectable levels of virus (HBV
DNA) and normal liver function (normal alanine aminotransferase or ALT).
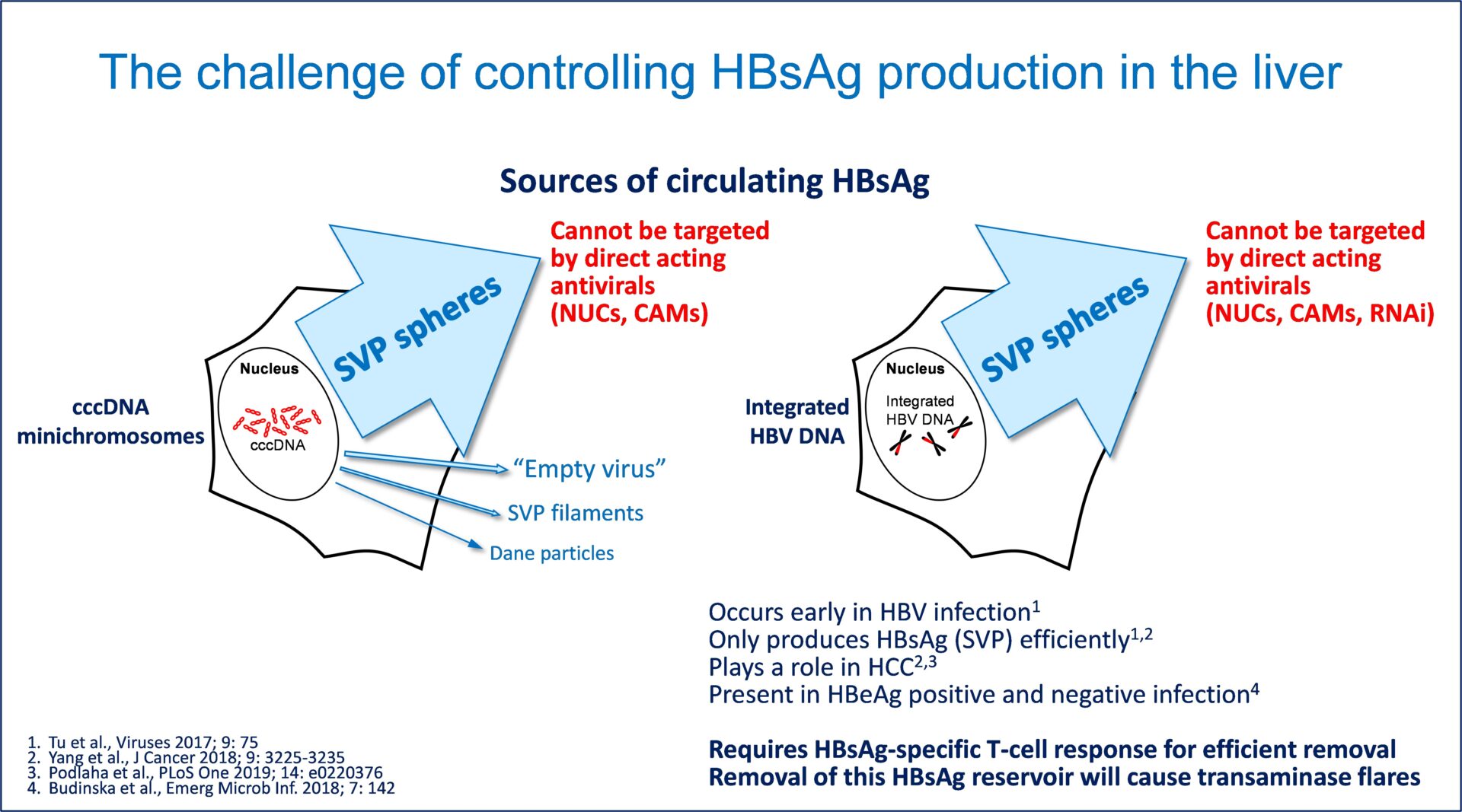
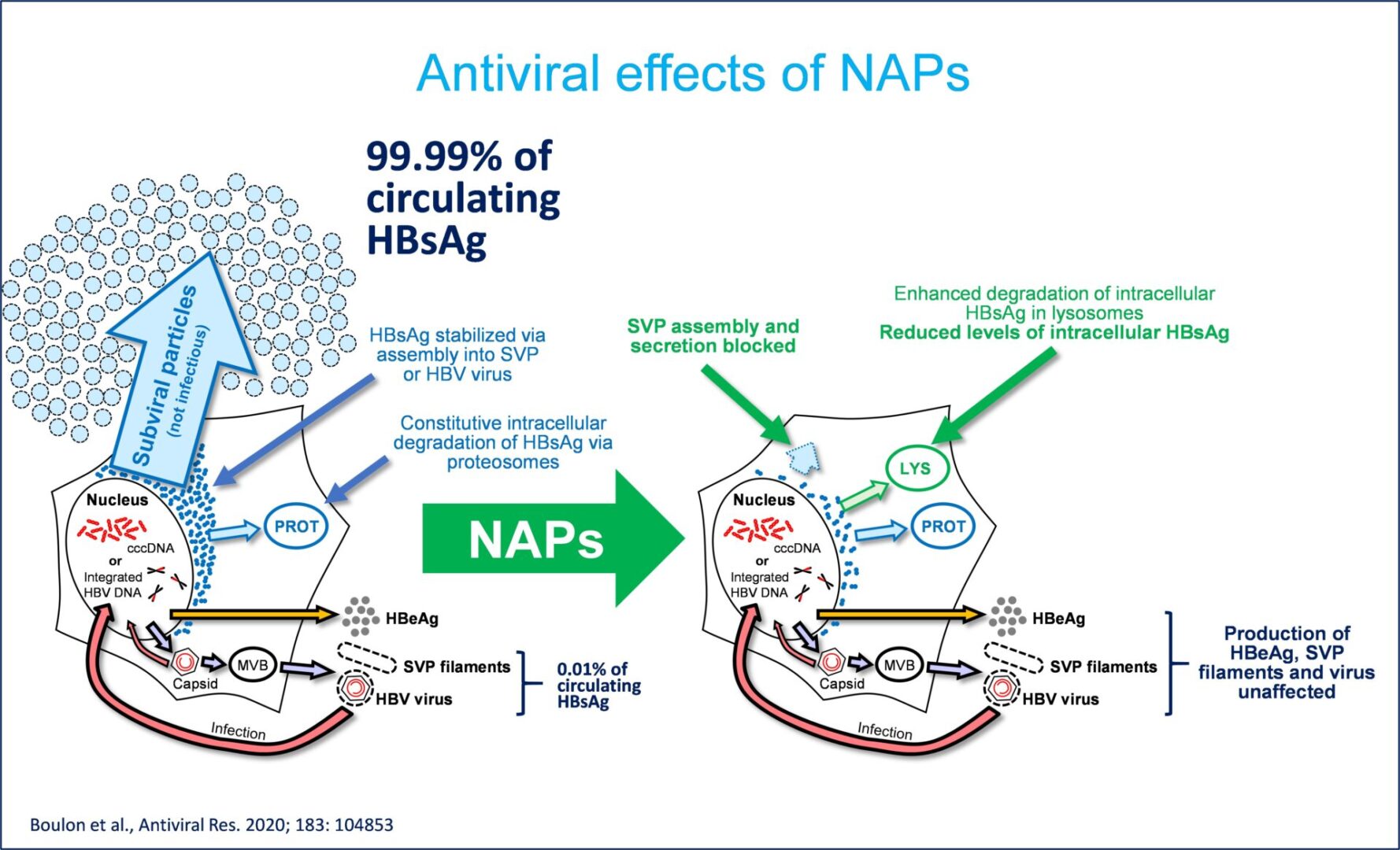
The major hurdle in achieving functional cure of HBV is the clearance of the HBsAg protein from the blood
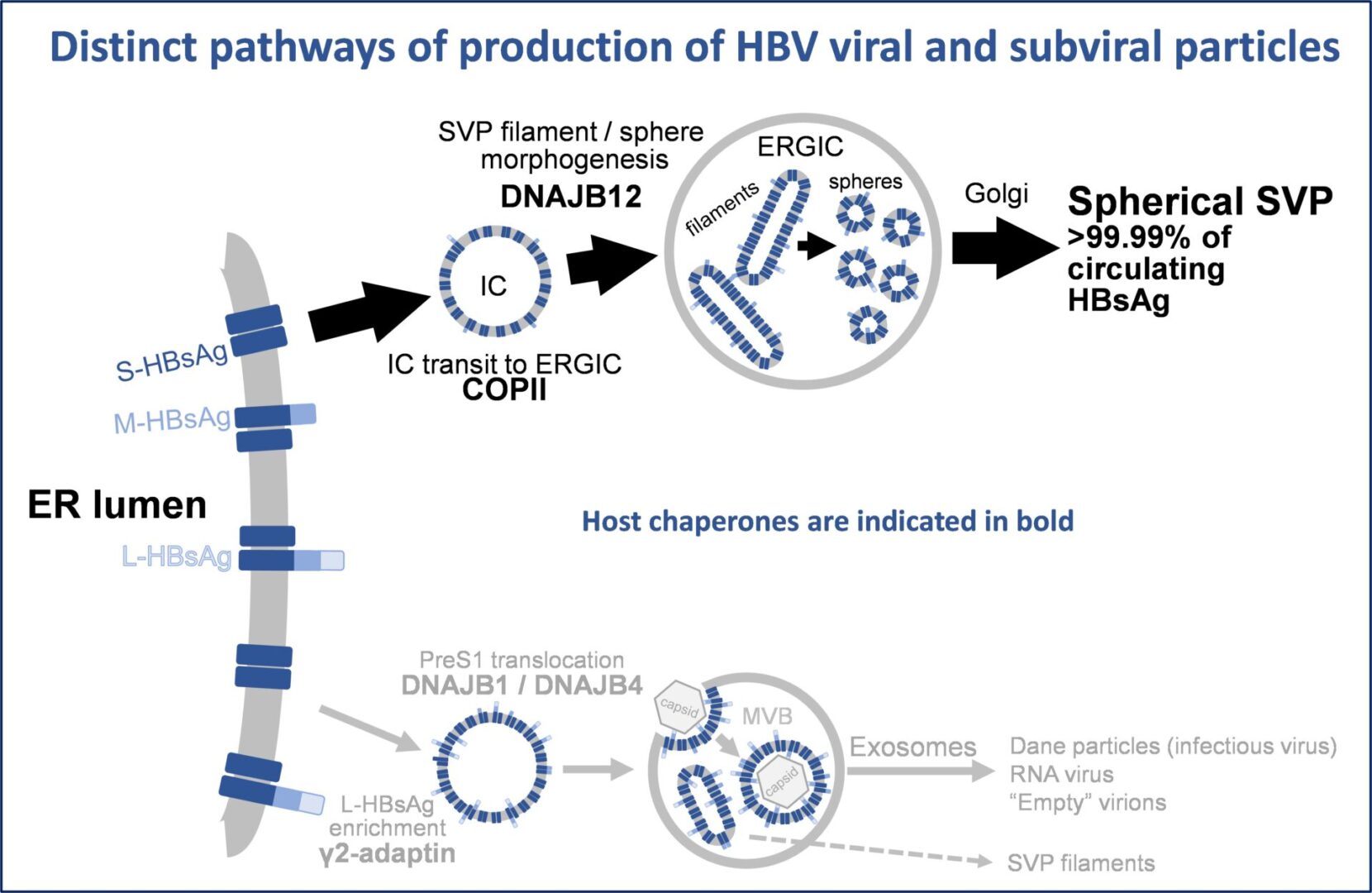
and the liver. This protein is by far the most abundant circulating viral antigen and is comprised almost entirely
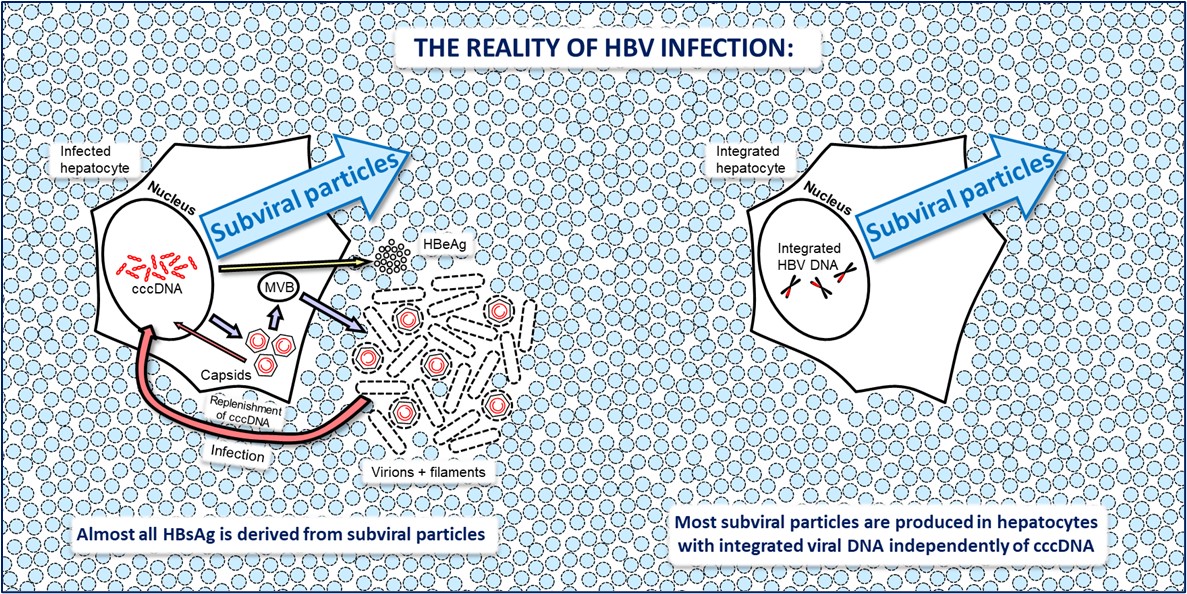
from non-infectious subviral particles (SVP). Not only are SVP produced by a mechanism independent from
viral production, a permanent source of SVP also exists in the HBV DNA integrated into host chromosomes,
which cannot make virus but can efficiently produce SVP.
Circulating SVP function to exhaust or inhibit important immune functions required to achieve functional cure
and also prevent immunotherapies from working properly. As such, clearance of SVP is a critical step in
achieving functional cure.
The problem with current treatments for HBV infection
Currently approved therapies for HBV either attempt to improve the immune function of the patient to control
the virus (interferons) or prevent the release of infectious virus by blocking the enzyme that is responsible for
the maturation of the virus into its infectious state (HBV reverse transcriptase inhibitors like entecavir [ETV]
and tenofovir disoproxil fumarate [TDF]). Both of these approaches suffer from important drawbacks: interferons can only restore control of infection that persists after therapy (“functional cure”) in a small number of patients (typically < 10%) primarily due to the persistent inhibitory effects of HBsAg. On the other hand, drugs like ETV and TDF work well to control the virus in most patients but cannot affect the production of SVP, rarely achieving functional cure when these medicines are stopped, making them a life-long treatment.
Agents that target the assembly of the viral capsid (capsid assembly modulators or CAMs) are also unable to
alter HBsAg levels and recent clinical data indicates these will not be able to surpass the antiviral effects of
ETV and TDF.
Antisense and RNAi also seek to target HBsAg production by targeting the degradation of HBV mRNA inside
hepatocytes. Numerous clinical trials have unfortunately demonstrated that the highly mutable nature of HBV
(it can exist in thousands of quasispecies in a single host) easily evades the primary mechanism of these
agents, with moderate 1-2 log reductions in HBsAg from baseline (still leaving abundant HBsAg present) likely
driven by off target immunostimulatory effects silencing cccDNA but not affecting integrated HBV DNA.
Nucleic acid polymers: unique agents directly targeting the assembly and secretion of HBV SVP
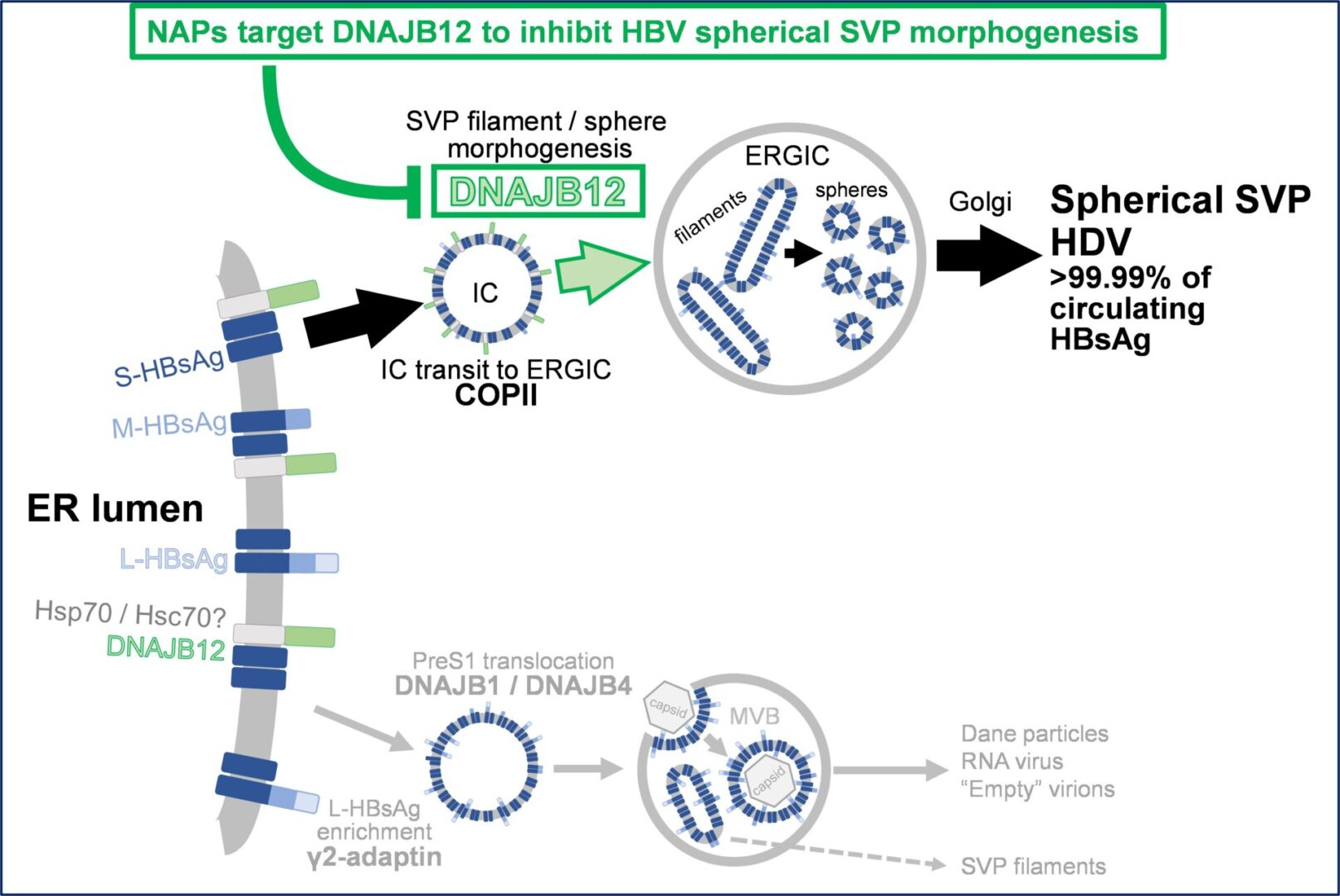
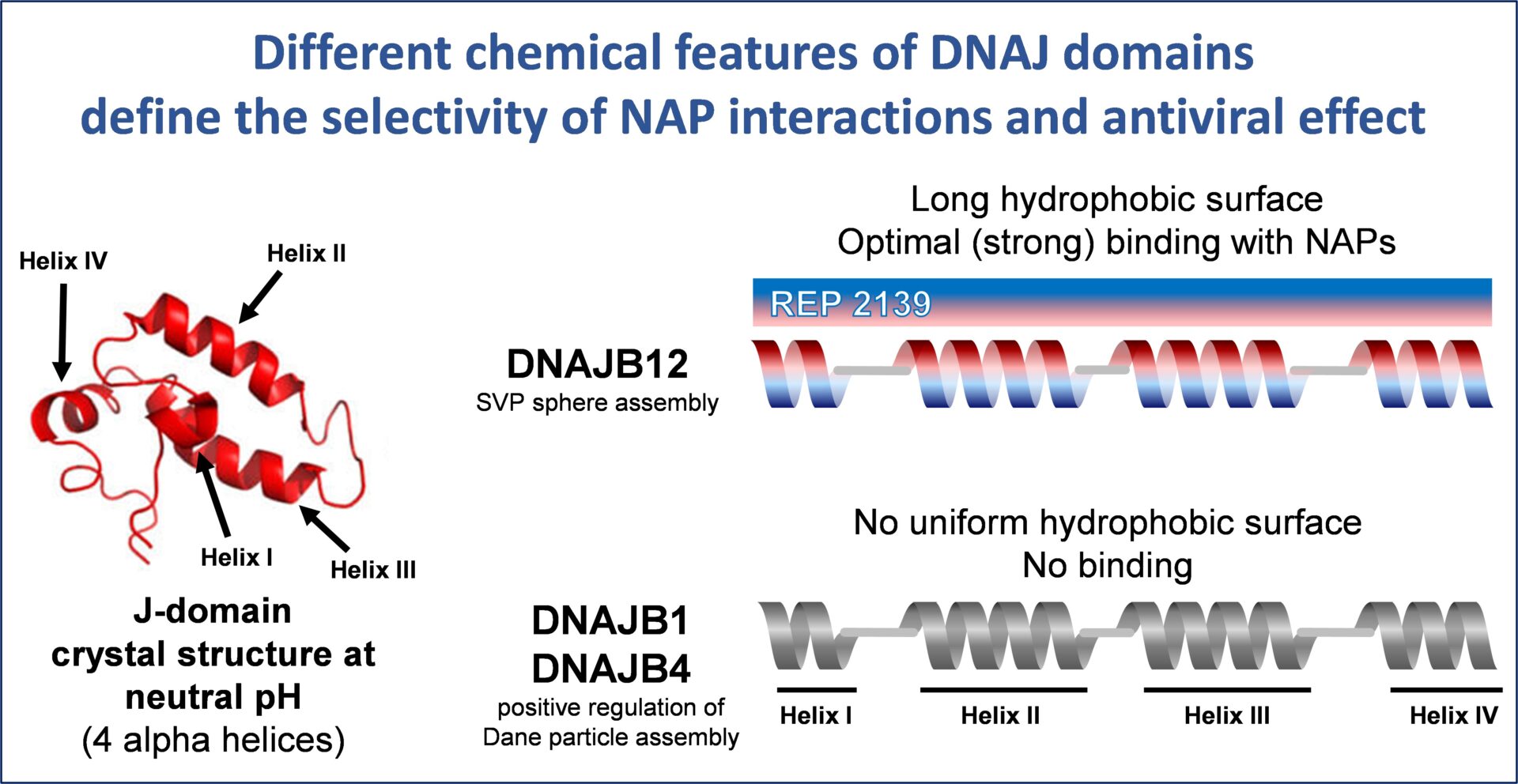
In HBV infection, NAPs have been shown to interact with the J-domain of the host HSP40 chaperone
DNAJB12 which is involved in spherical SVP assembly. The J-domain of DNAJB12 has four amphipathic
alpha helices which become exposed in the acidic lumen of post ER intermediate compartments. These
exposed alpha helices in DNAJB12 are selectively bound by NAPs, preventing the assembly of spherical
subviral particles. The amphipathic character of the alpha helices in the J-domain of DNAJB12 is unique,
driving the selective high affinity interaction of NAPs with DNAJB12 and not other members of the HSP40
family.
The selective inhibition of DNAJB12-mediated spherical HBV assembly by NAPs has two important antiviral
effects: 1) effectively block the replenishment of HBsAg in the circulation and 2) lower intracellular HBsAg via
constitutive degradation of the larger available pool of unassembled HBsAg dimers.
The clinical impact of REP 2139 when added to the existing standard of care
In pre-clinical and clinical studies in HBV infection, REP 2139 therapy by itself is accompanied by rapid
declines in HBsAg in the blood and liver. As a result of the removal of this immunosuppressive antigen burden,
many other antiviral responses accompany the primary effect of REP 2139 therapy and include:
- HBsAg seroconversion.
- HBeAg seroconversion (in HBeAg positive patients).
- Clearance of HBV DNA and HBV RNA (defective, non-infectious virus).
- Early inhibition of viral replication (cccDNA).
- Early reduction of HBV DNA and cccDNA in preclinical studies.
- Persistent control of HBV infection in the liver after removal of all therapy in preclinical studies (inactive
cccDNA, low or undetectable HBsAg) “functional cure”.
With the removal of HBsAg to levels below 1 IU/mL (as low as 0.005 IU/mL) occurring in most patients (typically
4-7 log reduction from baseline), the addition of different immunotherapies is accompanied by restoration of
immune control in humans which results in:
- The appearance of host mediated transaminase flares resulting from immune clearance of infected
hepatocytes from the liver. - The establishment of high rates of functional cure (39%) and virologic control (39%)
- Elimination of the need for further treatment in 78 % of patients.
These on-treatment and off-treatment results are unmatched by any other single drug or combination of drugs
either approved or in development.